A clinical trial led by University College London (UCL) and UCLH has revealed a potentially transformative approach to treating specific types of bowel cancer. By administering immunotherapy before surgery —rather than the traditional method of chemotherapy after surgery—researchers have observed patients remaining cancer-free for nearly three years.
A Shift in Treatment Strategy
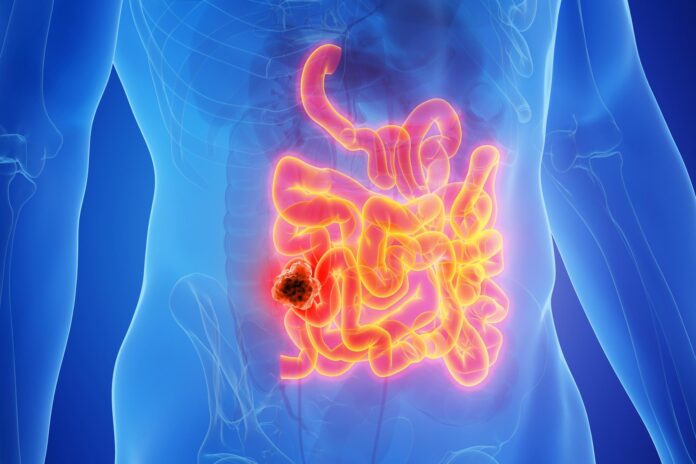
The findings from the NEOPRISM-CRC clinical trial, presented at the 2026 American Association for Cancer Research (AACR) Annual Meeting, focus on a specific subtype of cancer: MMR deficient/MSI-high bowel cancer. While this accounts for only 10–15% of bowel cancer cases, it represents a critical group where traditional treatments often struggle.
In this trial, patients with stage two or three cancer received a nine-week course of pembrolizumab (an immunotherapy drug) prior to their surgical procedure. The results have been striking:
- High Success Rate: 59% of patients showed no detectable cancer following the treatment and surgery.
- Durable Remission: After 33 months of follow-up, not a single patient in the study has experienced a recurrence.
- Comparison to Standard Care: Under current standard protocols (surgery followed by chemotherapy), approximately 25% of patients in this category are expected to relapse within three years.
Precision Medicine: Predicting Success via Blood Tests
Beyond just proving the treatment works, the study has opened doors to personalized oncology. Researchers have developed specialized blood tests designed to monitor “circulating tumor DNA.”
This allows doctors to move away from a “one size fits all” approach. By analyzing blood samples and immune profiling from tumor tissue, clinicians can now:
1. Detect early signs of success: If tumor DNA disappears from the blood, it is a strong indicator of long-term remission.
2. Tailor treatment intensity: Doctors can identify which patients are responding well and may require less aggressive follow-up, versus those at high risk who need more intensive intervention.
“What is particularly exciting is that we now may be able to predict who will respond to the treatment using personalized blood tests… This could help us identify patients who are doing well versus those who need additional treatment.” — Dr. Kai-Keen Shiu, Chief Investigator
The Human Impact: A Case Study in Recovery
The clinical data is mirrored by the experiences of trial participants. Christopher Burston, a 73-year-old patient diagnosed with stage 3 cancer, participated in the trial after a routine screening.
Following three doses of immunotherapy, his surgical team noted that the tumor had “essentially melted away.” Nearly three years later, Burston remains cancer-free and has returned to his normal daily activities, highlighting the potential for this method to significantly improve quality of life by reducing the need for heavy post-operative chemotherapy.
Why This Matters
Bowel cancer is the fourth most common cancer in the UK, with 44,000 new cases annually. While early detection (Stage 1) offers high survival rates, Stage 3 and Stage 4 diagnoses present much steeper challenges.
The NEOPRISM-CRC trial suggests that for the MSI-high subtype, preoperative immunotherapy may not just be an alternative, but a superior method for achieving long-term disease control and preventing the recurrence that often follows standard surgical intervention.
Conclusion: By utilizing immunotherapy before surgery and leveraging personalized blood tests to monitor progress, this new approach offers a highly effective, durable, and customizable pathway for patients with high-risk, specific genetic profiles of bowel cancer.