When actor Sam Neill recently revealed that his stage-three cancer was in remission following CAR T-cell therapy, he didn’t call it a miracle. He called it “science at its best.”
This sentiment echoes through the scientific community. While the treatment remains expensive and complex, experts describe it as a potential game-changer in oncology. For the first time, doctors are not just attacking cancer with external chemicals, but reprogramming the body’s own immune system to hunt down disease cells with precision.
“We’ve just scratched the surface on what’s possible,” says Professor Misty Jenkins, an immunologist at the Walter and Eliza Hall Institute.
Here is how this emerging technology works, why it is currently limited, and what the future holds for cancer treatment.
How It Works: Adding a ‘GPS’ to Immune Cells
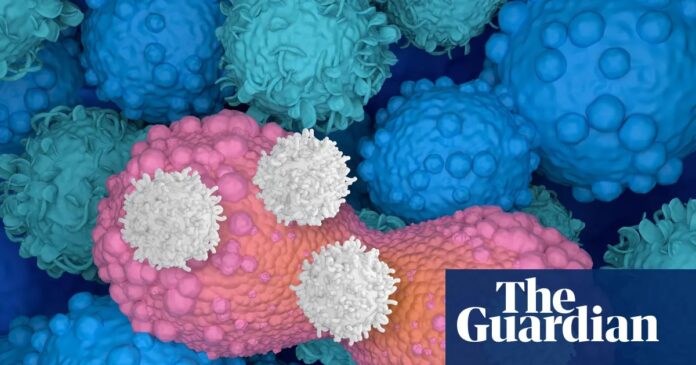
To understand CAR T-cell therapy, one must first understand the immune system’s natural defenders. The body uses white blood cells, specifically cytotoxic T-cells, to identify and destroy threats like bacteria, viruses, and cancerous cells. These cells act as mercenaries, locking onto invaders and destroying them.
However, cancer cells are masters of evasion. They often disguise themselves or suppress the immune response, allowing them to grow undetected.
CAR T-cell therapy (Chimeric Antigen Receptor T-cell) solves this by enhancing the T-cells’ targeting ability. The process involves three key steps:
- Extraction: Doctors filter a patient’s T-cells from their blood.
- Engineering: In a laboratory, these cells are genetically modified to produce a special receptor—a “GPS” that allows them to recognize specific proteins found only on cancer cells.
- Infusion: The supercharged T-cells are multiplied in the lab and infused back into the patient.
Once inside the body, these engineered cells hunt down and destroy cancer cells. Unlike chemotherapy, which attacks rapidly dividing cells indiscriminately (often causing severe side effects), CAR T-cell therapy is highly specific. It typically requires only one infusion and a few weeks of hospitalization, compared to months of traditional chemotherapy.
The ‘Living Drug’ Advantage
The most significant advantage of CAR T-cell therapy is its potential longevity. Traditional drugs leave the body after they are metabolized. CAR T-cells, however, are living drugs.
Dr. Criselle D’Souza, an immunologist at the Peter MacCallum Cancer Centre, explains that if the therapy successfully creates a “memory” in the immune system, the protective effect can persist indefinitely. This prevents relapse, a major challenge in cancer care.
A notable example is Emily Whitehead, the first child treated with CAR T-cell therapy in 2012. Her cancer has not returned, demonstrating the long-term potential of this approach. Since 2018, four CAR T-cell therapies have been approved in Australia, all currently focused on blood cancers such as leukemia and lymphoma.
The Solid Tumor Challenge
While success rates for blood cancers are high, extending this treatment to solid tumors (such as those in the breast, brain, or prostate) has proven difficult.
Associate Professor Maté Biro of the Garvan Institute compares the difference to the ease of the task:
* Blood cancers: Like “shooting fish in a barrel.” The cancer cells circulate freely, making them easy targets for T-cells.
* Solid tumors: Like “storming a formidable fortress.” These tumors are dense, often have poor blood supply, and create a microenvironment that suppresses T-cell activity.
Researchers are actively working to overcome these barriers. Strategies include:
* Enhancing T-cell weaponry: Engineering cells to secrete drugs or antibodies that help them survive in the hostile tumor environment.
* Improving targeting: Developing more precise “GPS” receptors to help T-cells penetrate the tumor structure.
Early trials have shown promise in prolonging the lives of patients with gastrointestinal cancers and pediatric brain tumors, but widespread application remains a work in progress.
The Future: Injectables and Lower Costs
Two major hurdles currently limit the accessibility of CAR T-cell therapy: cost and complexity.
- High Cost: Current treatments can exceed $500,000 AUD per patient. The manufacturing process is labor-intensive, requiring individualized lab work for each patient.
- Infrastructure: The therapy requires specialized hospital facilities to manufacture and administer the cells, which are not yet available in all regions.
However, the future may see a paradigm shift with in vivo CAR T-cell therapy. Instead of extracting cells and engineering them in a lab, patients could receive a single injection that instructs the body to produce CAR T-cells internally. This approach, similar to vaccination, could reduce costs by an order of magnitude and make the treatment more accessible.
A Call for Investment
Despite the high costs, governments are beginning to step in. Recently, the Australian federal government announced that Carvykti, a CAR T-cell therapy for multiple myeloma, would be funded in public hospitals, removing the financial barrier for eligible patients.
Nevertheless, experts warn that without sustained investment in local research and manufacturing capabilities, Australia risks falling behind.
“Science is delivering,” says Associate Professor Biro. “It’s the politicians and the health system that need to catch up.”
As CAR T-cell therapy evolves from a niche treatment for blood cancers to a broader weapon against solid tumors, the medical community remains cautiously optimistic. The technology is no longer just experimental; it is a proven, life-saving tool that is rapidly maturing. The next decade will determine whether it becomes a standard, accessible cure for millions or remains a luxury for the few.